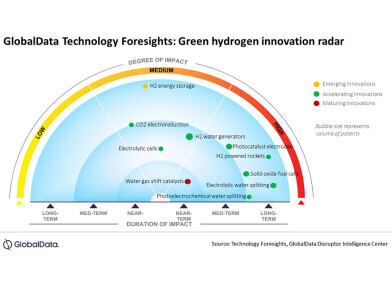
-
 Graphite structure
Graphite structure -
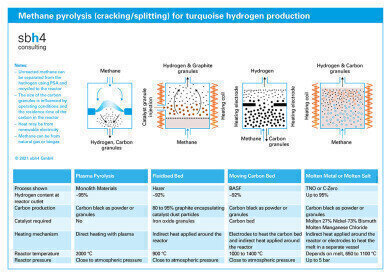 Methane pyrolysis for turquoise hydrogen production frame
Methane pyrolysis for turquoise hydrogen production frame
Green Energy
Technologies for turquoise hydrogen
Feb 23 2021
Author credit – Stephen B. Harrison, Managing Director, sbh4 consulting
Hydrogen must be produced at scale as a clean energy vector to mitigate greenhouse gas emissions and combat climate change. The use of carbon capture to mitigate the CO2 emissions from grey or black hydrogen production to result in blue or purple hydrogen is one option.
Green hydrogen produced on electrolysers fed with renewable electrical power is another. Turquoise hydrogen produced by methane splitting, also known as methane pyrolysis or cracking is also a potential pathway to low-carbon hydrogen.
Methane pyrolysis is endothermic, meaning that it requires heat energy to convert methane to hydrogen and solid carbon. There are different options for the heat supply. Indirect heating using burners fuelled by hydrogen or natural gas as a fuel is one option. Indirect electrical heating or direct heating with an electrical plasma are also possible.
Research into methane pyrolysis has been undertaken since the 1960’s. In the past 10 years methane pyrolysis has picked up momentum and several companies have piloted various technologies.
Monolith Materials started development of their methane pyrolysis process in 2012. Methane is heated by means of renewable electricity to 2,000 °C. At this temperature, the methane molecule splits and forms a plasma. This leads to the formation of carbon black, while the protons split off from the methane molecule and recombine to form hydrogen molecules.
From 2013-2017, a lab-scale reactor was built and operated by BASF at Ludwigshafen, Germany. It identified key process parameters and construction of a larger pilot plant soon followed. Both projects were supported by the BMBF (German Federal Ministry of Education and Research).
In the BASF process, external electrical heating to 1000 °C is used to drive the pyrolysis reaction. Since the heating is electrical, the process could use renewable energy. The reactor vessel is a vertical cylinder. Methane enters at the base and flows upwards through a downward-moving bed of solid carbon granules. The methane decomposes to form carbon that binds to the bed of carbon granules and hydrogen gas. Hydrogen and unreacted methane rise to flow from the top of the reactor vessel. Solid carbon leaves from the lower part of the column.
Hazer has operated a fluidised bed methane pyrolysis reactor pilot plant in Kwinana, Western Australia since 2016. Work has commenced on planning a commercial-scale demonstration plant. In the Hazer process, a fluidised bed reactor is externally heated to 900 °C. Iron oxide powder is injected to act as a catalyst. Methane gas is introduced from the bottom of the reactor. As the methane molecule splits, carbon atoms form graphite on the catalyst surface and hydrogen is released. The upward flow of hot gases carries the low-density graphite particles out of the top of the reactor.
In molten metal methane pyrolysis, methane gas is bubbled upwards through a column of molten metal which is a catalyst to convert methane to carbon and hydrogen. The process is being considered by some Russian natural gas producers, such as Gazprom, for turquoise hydrogen production. A challenge of the molten metal bubbler reactor is separation of the solid carbon from the molten metal at the top of the reactor. TNO has innovated a solution to this issue by floating a layer of molten salt on top of the molten metal.
Other researchers have resolved the problem by replacing the molten metal with a molten salt in the entire bubble column. In California, C-Zero has experimented with this process and has determined that manganese chloride is a particularly effective catalyst for methane splitting.
A question that arises from methane pyrolysis with hydrogen as the target is: what happens with the various forms of solid carbon that are produced? If turquoise hydrogen production becomes a mainstream pathway to hydrogen, the amount of solid carbon produced will greatly exceed demand from current applications. If carbon black becomes abundant at low cost, it might find additional application as a soil-improver in agriculture or as a substitute for coke in ironmaking.
Turquoise hydrogen can be produced from renewable biomethane and renewable electrical power. It therefore has the potential to be part of the green energy revolution.
Events
Apr 22 2024 Hannover, Germany
Apr 23 2024 Kuala Lumpur, Malaysia
Apr 24 2024 Sao Paulo, Brasil
May 05 2024 Seville, Spain
May 13 2024 Munich, Germany