Aquaculture is the farming and harvesting of aquatic organisms within a controlled environment. The aquaculture sector is now the fastest growing animal food sector, with approximately 46% of all fish consumed worldwide in 2012 being produced within aquaculture facilities. In accordance with the UN FAO report; State of World Fisheries and Aquaculture 2012 (SOFIA), aquaculture production reached an estimated total value of US$125billion in 2010, expanding at an average annual rate of 8.8% over the last 3 decades, with this figure set to rise further over the coming years.1 Maintaining water quality and stabilising water parameters is one of the biggest concerns facing the aquaculture industry, with a healthy aquatic environment essential to the success of the operation. Whilst the fish are kept within a controlled environment, the use of ponds and recirculation systems provides an extra challenge, especially with the intense, closed nature of the cultures.
Of all the water quality parameters that affect and influence fish behaviour and health, ammonia is one of the most important. Ammonia is a colourless gas compound of nitrogen and hydrogen, with a strong pungent smell at increased concentrations. Within an aquatic environment, ammonia occurs in two forms: Ionised ammonia is relatively non-toxic and has the chemical formula NH4+, the toxic un-ionised form of ammonia has no charge and has the chemical formula NH3.2 The form of ammonia present at any given time is dictated by the water temperature and the pH, with rising alkaline conditions often preceding an increase in the toxic un-ionised ammonia levels.3
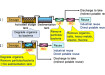
Ammonia plays an important part within the nitrogen cycle of any aquatic environment. This is the oxidative process in which ammonia is first converted into nitrite (NO2) by naturally occurring Nitrosospira and Nitrosomonas bacteria in the water, before further bacterial species Nitrospira and Nitrobacter convert the nitrite into nitrate (NO3). This nitrification process occurs either on the surface of the mud substrate and plants or within the biofilter of a tank based system. The nitrite is still toxic to fish species but encourages the growth and colonisation of Nitrobacter to convert it to the less toxic nitrate form. The nitrate is then taken up by aquatic plants and algae in aquaculture systems. The nitrogen cycle is directly influenced by oxygen and alkalinity levels, with a decrease in either terminating the cycle and thus increasing ammonia and nitrite levels once more.4 It is worth noting that the ammonia levels favour the growth of specific phytoplankton and algal species, which alters the aquatic biodiversity and dynamics of an ecosystem.
Ammonia enters the aquatic environment primarily from the fish themselves via their waste. Fish diet will often contain high levels of protein, which when metabolised, produces ammonia as a by-product. The fish then excretes the ammonia from its blood into the water via a partial pressure gradient across its gill epithelium, as well as a small amount in the urine.5 The dietary protein concentration in the feed is directly proportional to the ammonia levels produced, with higher protein levels producing more ammonia. Other sources can include the decomposition of algae or uneaten food in the aquatic environment, with smaller volumes of water more susceptible to ammonia spikes.
Ammonia ‘sinks’ occur within pond aquaculture in the form of the plants and algae. These organisms require nitrogen as a key nutrient for growth, and as a result absorb ammonia from the water. As rates of photosynthesis increase, plant and algal growth increases and the uptake of ammonia increases. Whilst the presence of algae within an aquaculture system does directly cause a reduction in ammonia levels, it is not a viable long-term method for ammonia removal. During the winter months, ammonia production is consistent but algal ammonia uptake is limited. This causes an increase in ammonia levels, and consequently fish may become stressed at a period of time during which the immune system of a fish is suppressed due to lower temperatures.9 Algal blooms are also subject to crashing, whereby the algae population, without explanation, dies suddenly. In such instances, the algal matter reverts from a sink to a source of ammonia, as well as reducing the dissolved oxygen concentrations and pH and increasing the levels of carbon dioxide present.
Ammonia affects the fish both directly and indirectly depending on the levels present, with certain species more susceptible to ammonia toxicity than others. At lower concentrations, around 0.05mg/L, un-ionised ammonia is harmful to fish species and can result in poor growth and feed conversion rates, reduced fecundity and fertility and increase stress and susceptibility to bacterial infections and diseases.6 At higher concentrations, exceeding 2.0mg/L, ammonia causes gill and tissue damage, extreme lethargy and death. In winter when reduced feeds are administered, ammonia levels can be higher because the decrease in temperature reduces the rate of algal photosynthesis so less ammonia is removed by this means.
Tolerance to ammonia varies between species. The African lungfish (Protopterus dolloi) is capable of detoxifying ammonia into alternative waste products such as urea or glutamine,7 whilst species such as the giant mudskipper (Periophthalmodon schlosseri) reduce their ammonia with a partial amino acid catabolism.8 Other species such as the oriental weather loach (Misgurnus anguillicaudatus) are tolerant to more extreme ammonia concentrations.
Measuring the levels of ammonia within a body of water only provides a snapshot in time, and does not account for fluctuating levels during the nitrification process. When ammonia concentrations reach higher, compromising levels, there are a number of commonly proposed alternatives to rectify the issue. The effectiveness of each method is however open to debate and most are not long-term solutions.
Excess feed is one of the primary sources of ammonia build up, but reducing feeding rates is not a short-term solution and will have little immediate effect. In order to reduce the risks in the long-term and to minimise the associated effects of sub-lethal ammonia exposure, a managed conservative feeding rate with monitored protein content should be implemented, and adjusted seasonally. Feeding during periods of stress will further exacerbate the issue, as food will remain uneaten and contribute to elevated ammonia levels.
The addition of lime or phosphorus is another option. Adding a liming agent can work to correct extreme pH values, and is often implemented during late afternoon when the toxic form is likely to be at its highest.10 This method does not act to remove the ammonia, but rather alters the ammonia form present from toxic to non-toxic in low pH conditions. In fact, the addition of lime into ponds with adequate alkalinity may cause a rapid pH shift and only serve to compound the ammonia issue. Phosphorus acts as a fertiliser for algae, increasing the algal population and in consequence the uptake of ammonia.11 In aquaculture however, light is the main catalyst for algal blooms rather than nutrient availability so whilst useful when re-establishing an algal bloom after a crash, adding phosphorus to established algae population has little effect.
Other solutions include increasing aeration within a pond and flushing with freshwater. By aerating a pond, dissolved ammonia in the toxic form will diffuse from the water into the air. This works on a small scale but would be ineffective within larger aquaculture ponds, whereby increasing aeration may only serve to stir the benthic sediment and thus increase the ammonia levels further.12 Flushing through with freshwater is effective on a smaller scale, diluting and reducing ammonia levels present. However, within larger aquaculture operations, the volume of water required to reduced an ammonia spike is vast, and as a result, comes with economic and time costs and has the potential to release pond effluent into the local environment.
Within an aquaculture environment, particularly on a larger scale, there is no fast acting remedy to elevated ammonia levels, and it is the prevention rather than the correction that should be the focus, with regular monitoring a key component. Within smaller scale facilities with fewer aquaculture ponds or tanks, manual ammonia testing kits will suffice, with many different liquid or powder kits available on the market. These kits are simple to follow and provide a snap-shot ammonia reading. The use of a submersible in-tank product such as Seneye, which continuously monitors a variety of water parameters including ammonia, provides an accurate and more detailed record and works as an early warning system. On a larger scale, manual testing kits become time consuming, and the results may only fit within a range rather than provide an accurate figure. In this situation, handheld monitoring instruments are the best solution, allowing for efficient monitoring of ammonia levels across a larger number of holding pond or tank facilities. This reduces the time taken to monitor each individual water sample and to gauge the actual reading.
Fish growth is not influenced by brief daily exposures to elevated ammonia levels. Whilst it is apparent that there is no definitive way of reducing elevated ammonia levels in the short term, the focus should be on prevention. By undertaking continuous or regular monitoring of water quality parameters, daily and seasonal fluctuations and trends can be identified. This will then allow for better aquaculture management practices with regard to stocking and harvesting densities and feeding regimes to be implemented.
References
1. FAO. (2012). The state of world fisheries and aquaculture. Rome 2012.
2. Hargreaves, J.A., & Tucker, C.S. (2004). Managing ammonia in fish ponds. Southern Regional Aquaculture Centre.
3. Resdi, R., Jessy, R.S., & Ibrahim, M.H. (2013). Vermicast from cattle/goat manure as fish pond fertiliser and effect on water quality. Journal of Environmental Science, Computer Science and Engineering & Technology, 2(2): 234-240.
4. YSI. (2010). Understanding ammonia in aquaculture ponds. http://www.ysi.com/media/pdfs/ A585-Understanding-Ammonia-in-Aquaculture-Ponds.pdf.
5. Ip, Y. K., & Chew, S. F. (2010). Ammonia production, excretion, toxicity, and defence in fish: a review. Frontiers in physiology, 1:134.
6. Hargreaves, J.A., & Tucker, C.S. (2004). Managing ammonia in fish ponds. Southern Regional Aquaculture Centre.
7. Chew, S.F., Ong, T.F., Ho, L., Tam, W.L., Loong, A.M., Hiong, K.C., Wong, W.P., & Ip, Y.K. (2003). Urea synthesis in the African lungfish Protopterus dolloi; hepatic carbamoyl phosphate synthetase III and glutamine synthetase are upregulated by 6 days of aerial exposure. Journal of Experimental Biology, 206: 3615-3624.
8. Randall, D. J., Ip, Y. K., Chew, S. F., & Wilson, J. M. (2004). Air Breathing and Ammonia Excretion in the Giant Mudskipper, Periophthalmodon schlosseri*. Physiological and biochemical Zoology, 77(5):783-788.
9. Le Morvan, C., Troutaud, D., & Deschaux, P. (1998). Differential effects of temperature on specific and nonspecific immune defences in fish. The Journal of Experimental Biology, 201:165-168.
10. Boyd, C.E., & Massaut, L. (1999). Risks associated with the use of chemicals in pond aquaculture. Aquacultural Engineering, 20:113-132.
11. Crab, R., Avnimelech, Y., Defoirdt, T., Bossier, P., & Verstraete, W. (2007). Nitrogen removal techniques in aquaculture for a sustainable production. Aquaculture, 270:1-14.
12. YSI. (2010). Understanding ammonia in aquaculture ponds. http://www.ysi.com/media/pdfs/ A585-Understanding-Ammonia-in-Aquaculture-Ponds.pdf.